How Artificial Intelligence (AI) and Deep Learning (DL) processes are shaping and influencing the work of the experimental pathologist.
Introduction
Technology developments have changed and will continue to change many fields. When it comes to science and precise data, technology developments are becoming a real challenge for professionals such as pathologists who are used to certain work frames. Glass-stained slides have been the source material for the pathologist to gain information and data using microscopes, observational skills, experience and medical knowledge. The experimental pathologists can observe tissue changes and reflect those to the scientist who is confronted with scientific questions and hypotheses. Experimental pathologists involved in research of tissue samples from laboratory animals serve the academic world and the industrial platform of new drug and medical device development. Their way of reading and interpreting slides differs dramatically from colleagues who are reading slides for medical diagnosis.
This commentary is focused on the work patterns of those pathologists who are assessing animal study histology for the purpose of new pharmaceutical development and how the latest technological advances can influence their work frame.
The historical task of the pathologist in the pharmaceutical industry has been to assure that product candidates are fully assessed for any harm to normal tissues and that medical devices are evaluated for tolerability as invasive devices and in procedures. Animal studies that provide data showing satisfactory drug tolerability give the developer the comfort to proceed with product development for initiating and continuing clinical trials, hopefully for eventual regulatory approval (FDA, EMEA) as a commercial product. This approach by pathologists, called toxicological pathology, became an important branch in the repertoire of experimental pathology tasks, encompassing the knowledge tissue responses to different drugs as documented in specialized journals.
A second branch for pathologists has been efficacy studies in animal models that are employed to evaluate the potential of a drug to improve a condition or pathological process comparable to that in the target human disease. Animal models that reflect the human disease are created by transgenic technology or chemical, surgical or behavioural applications. Pathologists evaluate drug activity using different model parameter endpoints that are relevant for the target disease and pathology. Histological features are evaluated in animal efficacy study for current and candidate drugs and in different doses and regimens and if applicable in combination with other drugs relative to relevant negative and positive control groups. The validation of an animal model for relevance to human disease is a very complex and challenging process for academic and contract research institutes such as Jackson Laboratories.
The bias problem among pathologists
Bias has been of much concern to pathologists, each of whom has strengths and weaknesses in experience and knowledge of different tissues, tasks and techniques, which lead to subjectivity in the interpretation of results. To improve objectivity, tools have been developed such as second opinions and peer reviews by additional pathologists. Such bias has been less of a problem in toxicological safety studies, where the tolerance of tissue lesions has been very low. On the other hand, bias has been a bigger challenge for animal model efficacy studies. Comparing a new drug with a licenced drug could show minimal differences and inaccuracies by histology, which is partially subjective, thus leading to misinterpretations of data. Thus, it has been essential to develop quantitative and more accurate techniques such as LCMS/MS, HPLC and ELISA for evaluating tissues, blood and other fluids. Moreover, surveys have indicated the importance of microscopy image analysis for biological research.
Digital Image Processing (DIP), Deep Learning (DL) and digital slides interpretations.
New tools have been developed to extract objective information from images of complex structures such as cells and tissues. Besides traditional glass histological slides, digital slides transform scans of whole glass slides into high-resolution digital presentations of cells or tissues. Whole Slide Imaging (WSI) provides the pathologist a large database that can be analysed using digital tools such as morphometric operations, colour-based segmentation, colour deconvolution, thresholding, and clustering. These digital tools provide substantial qualitative and quantitative benefits in assessing pharmacologic effects and drug efficacy. These tools enable the pathologist to work accurately in visualizing data for characterizing relevant tissues and organelles and measuring structures such as alveoli or myofibers to identify different pathologies. Digital tools also enable pathologists to accurately and objectively extract data from tissues without bias or subjectivity, unlike the semi-quantitative method of lesion evaluation using grades and scores. Using the proper tool and stain, automatic quantifications and standardizations can be made, e.g., cell count, degree of fibrosis or myelinated fibers, and analysis of relevant tissue regions. Another rapidly developing field is Deep Learning (DL) for performing pattern recognition of histological components such as inflammatory cells, necrotic tissue, epithelium layers, blood vessels, cell membranes and organelles, collagen fibers, and organ components. The system is a paperless archive that can be automated into a ‘digital pathologist’ for identifying pathological features such as necrosis, oedema, inflammation neoplasia, atrophy and hypertrophy.
Example. Morphometric quantification of the amount fibrous tissue of diaphragm striated muscle in a MDX mouse model for muscular dystrophy as a tool for drug development.
The computation of relative fibrosis area seen in S stain slides (Figure 1) is followed by conversion to a digital slide (Figure 2) by MATLAB Color-Based Segmentation using the L*a*b* Color Space derived from the CIE XYZ tristimulus values and consisting of a luminosity ‘L*’ or brightness layer, chromaticity layer ‘a*’ indicating where color falls along the red-green axis, and chromaticity layer ‘b*’ indicating where the color falls along the blue-yellow axis. We chose a small sample region for each tissue and used these color markers to classify each pixel. The relative area was calculated as the (fibrosis area) / (muscle+fibrosis area).
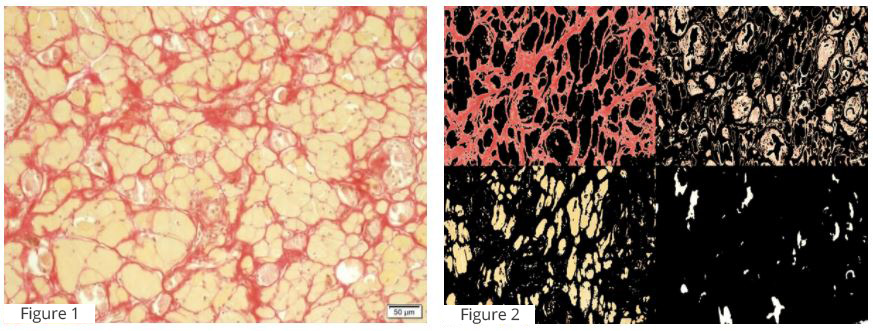

Figure 1. Diaphragm striated muscle. Slide of affected striated muscle stained with Serius Red (S stain), which shows amount of fibrous tissue between atrophic muscle fibers (yellow).
Figure 2. Diaphragm striated muscle. Digital slide showing tissue segmentation done by MATLAB Color-Based Segmentation using the L*a*b* Color Space.
Impact of histology slide digitalization, data processing & analysis on the pathologist’s work
These advanced digital tools enable the pathologist to evaluate cells and tissues more accurately and objectively. The expertise of the pathologist is needed for excellence in study planning, e.g., selecting the most appropriate animal model, choosing the best histology end points, optimizing the technical approach, and of course evaluating the specimen. The pathologist plays a central role in protocol design and study termination, including the tissue harvesting process. This planning also includes selecting staining types, relevant IHC markers and other histological tools (e.g., ‘free floating sections’ or plastic embedding sections), and assuring that technical aspects are optimized, including sample quality and slide grade and number per animal or organ. In the final phase of sample evaluation and interpretation, the pathologist controls the automated digital system, compares representative slides, and probes for deviated results or possible errors of digital slides relative to the glass slide. The pathologist also has a key role in the interpretation of histology results, using pathological and medical knowledge and experience to place the results in the proper context. Moreover, the advanced digital tools provide for a high level of quality control that is very important for regulatory and clinical filings and for due diligence reviews to support fund-raising and strategic collaborations with pharmaceutical development partners.
In summary we strongly believe that the introduction of the digital slide is a great opportunity of great benefit to the pathology discipline in general. We also think that this technology will improve our field of expertise by reducing bias and assuring more accurate results. This will improve the role of histology in efficacy studies compared to other measurement methods. Furthermore, the DL process will shed light on new pathological features and anatomical structures (e.g., the recently discovered glymphatic system). Furthermore digital slide image analysis will free the time od the pathologist, allowing him / her to have more time to focus on understanding pathogenesis, study designs, checking accuracy of the scientist work and hypetheses, instead of spending the all day counting cells.

